Science & Tech
Humans Just Grew “Synthetic” Embryos in a Lab Without Using Sperm or Eggs for the First Time in History
However, the experiment raises serious ethical questions.
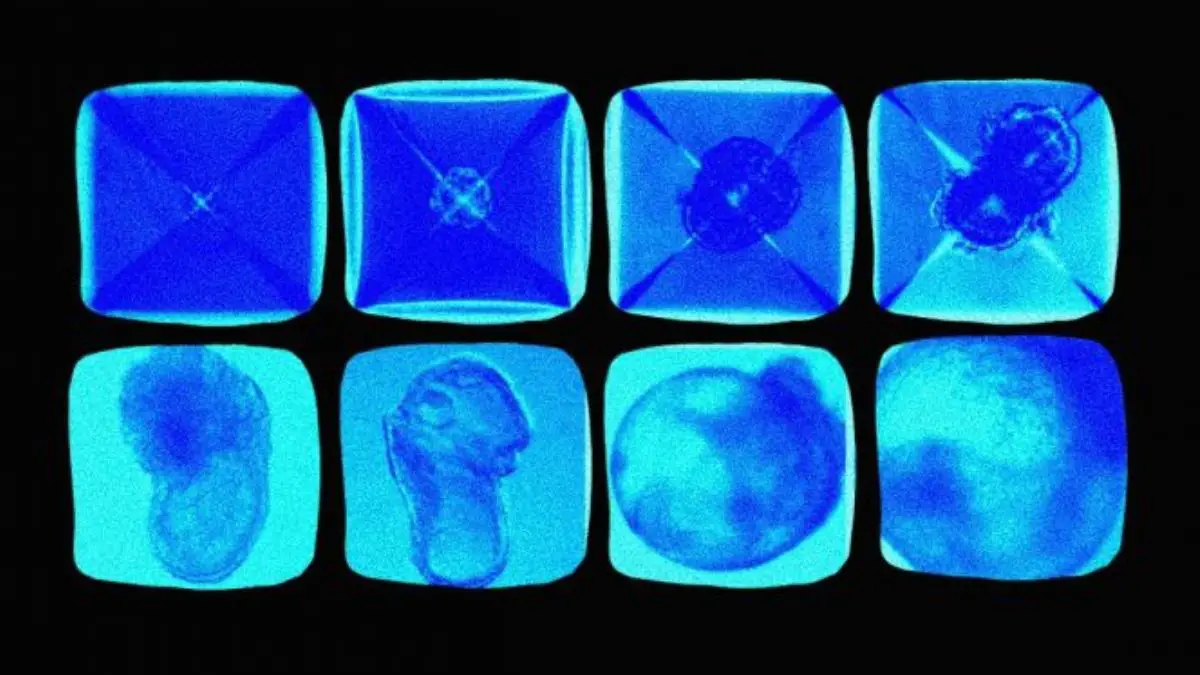
A team of stem cell scientists at the Weizmann Institute in Israel have announced the creation of the world’s first-ever “synthetic” embryos in a groundbreaking feat that bypassed the need for sperm, eggs and fertilization.
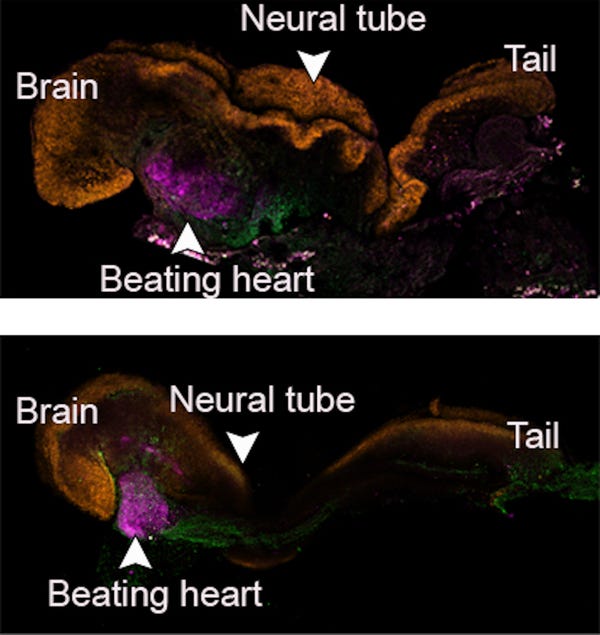
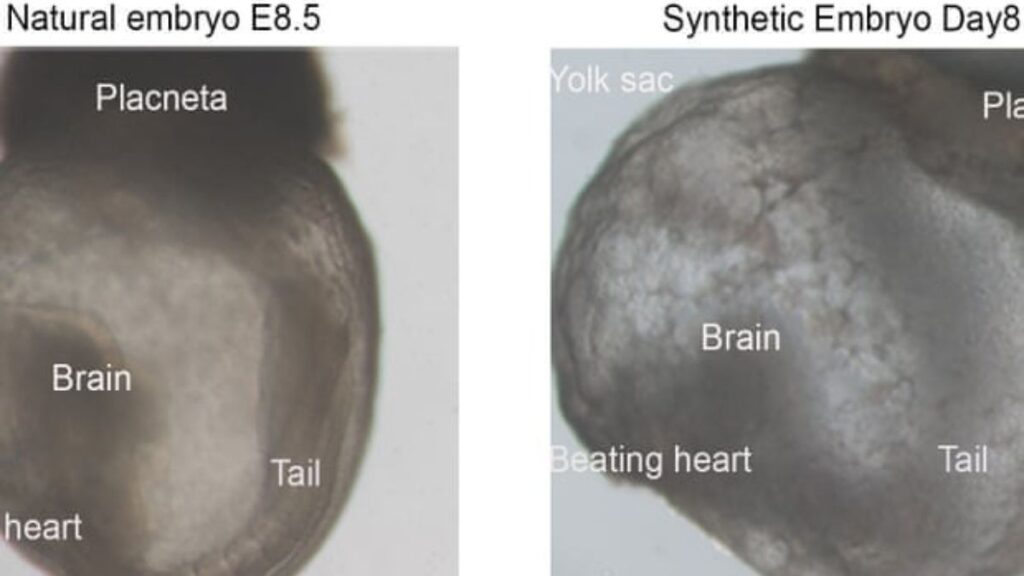
The researchers discovered that it was possible to get stem cells from mice that were grown into an artificial womb for eight days to self-assemble into “synthetic” embryos that had an intestinal tract, a developing brain, and a beating heart.
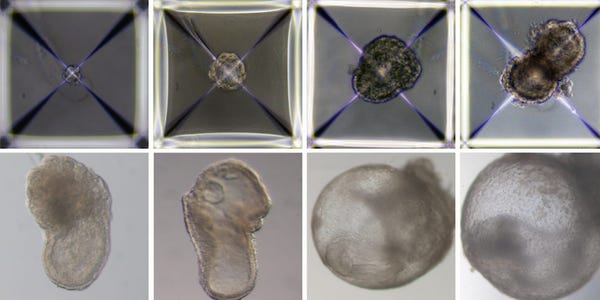


The embryo stopped growing after eight days, which is the equivalent of three months of pregnancy for a mouse.
“Remarkably, we show that embryonic stem cells generate whole synthetic embryos, meaning this includes the placenta and yolk sac surrounding the embryo,” said Professor Jacob Hanna, who led the research.
“We are truly excited about this work and its implications.”
The team’s remarkable work, a world-first has been published in peer-reviewed journal Cell on August 1, 2022.
However, the research also raises serious ethical questions which leave many people uncomfortable.
Have we stopped to contemplate boundaries as stem cell research continues to advance and become more successful, enabling scientists to create human organs and even embryos from stem cells?
The International Society for Stem Cell Research (ISSCR) released new guidelines last year limiting the culture of human embryos in the laboratory to 14 days, before the first indications of the nervous system start showing, in an effort to address this question.
After 14 days, the embryos must then be destroyed.
Dr. James Briscoe, a principal group leader at the Francis Crick Institute in London, said it was crucial to discuss how to regulate the work before human synthetic embryos were developed.
“Synthetic human embryos are not an immediate prospect. We know less about human embryos than mouse embryos and the inefficiency of the mouse synthetic embryos suggests that translating the findings to human requires further development,” Briscoe said.
“Now is a good time to consider the best legal and ethical framework to regulate research and use of human synthetic embryos and to update the current regulations,” he added.
Human embryos are already subject to regulation, but artificial human embryos are a novel concept. One day, could a person’s womb be used to implant a synthetic human embryo?
According to researchers at the Weizmann Institute, synthetic mouse embryos cannot be regarded as being the same as natural mouse embryos, and so far all attempts to implant them into the womb of a mouse did not result in a pregnancy.
The Weizmann Institute experts believe the results of the experiment could hold the key to understanding how organs develop in the embryos and eventually creating replacement organs for people who require transplants.
The race to create the best synthetic embryo from human and mouse stem cells is underway.
The same team reported last year how they had created an artificial womb that allowed natural mouse embryos to develop outside the uterus for a few days. In the latest research, the same apparatus was used to grow mouse stem cells for more than a week—nearly half the duration of a mouse’s gestation.
Professor Paul Tesar, a geneticist at Case Western Reserve University, said in an interview with StatNews that as scientists advance stem cell-derived embryos farther and further down the course of development, the more the synthetic and natural embryos increasingly converge.
“There will always be a grey area,” Tesar said. “But as scientists and as a society we need to come together to decide where the line is and define what is ethically acceptable.”
The scientists from the Weizmann Institute claimed their experiment’s only goal was to prove whether it could one day be feasible to develop synthetic embryos able to grow replacement organs for those in need of transplants.
“Our goal is not making pregnancy outside the uterus, whether it’s mice or any species,” lead researcher Dr. Jacob Hanna told the Washington Post.
“We are really facing difficulties making organs—and in order to make stem cells become organs, we need to learn how the embryo does that. We started with this because the uterus is a black box—it is not transparent”.
Typos, corrections and/or news tips? Email us at Contact@TheMindUnleashed.com
